
The Pfizer/BioNTech COVID-19 vaccine has been provisionally approved for use in Australia by the Therapeutic Goods Administration (TGA).
Subscribe now for unlimited access.
$0/
(min cost $0)
or signup to continue reading
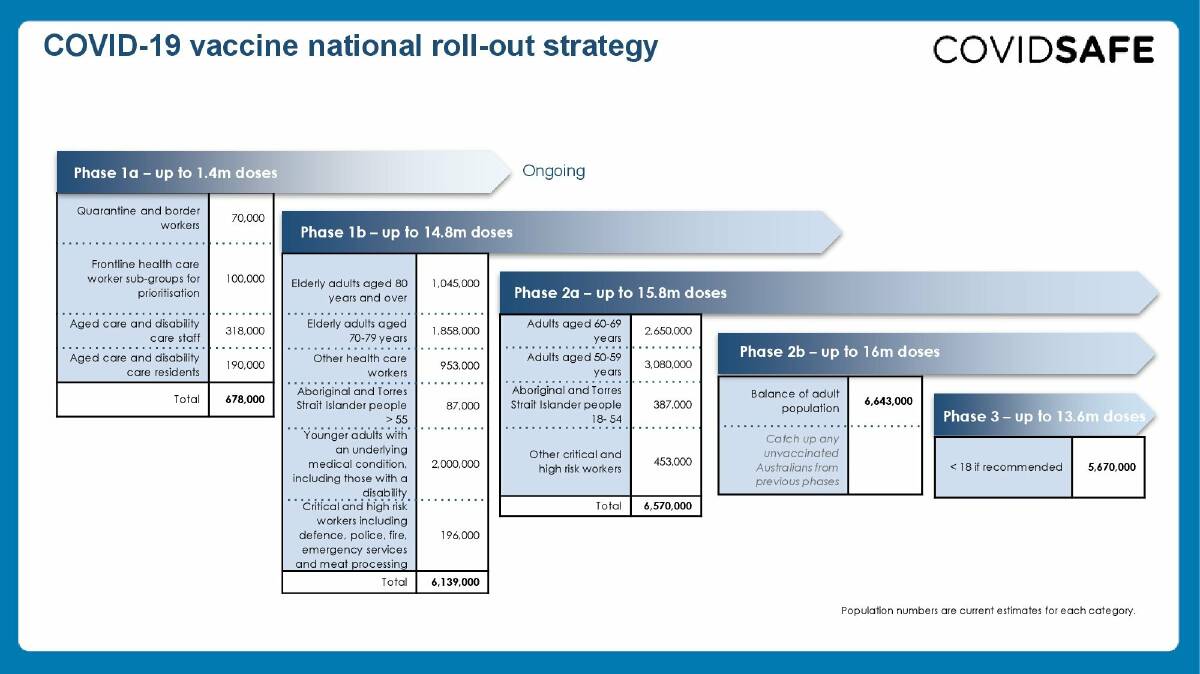
The vaccine will be provided free to Australians and is set to be rolled out in five strategic phases, with a priority group of Australians expected to receive their first dose of the COVID-19 vaccine in late February to early March.
Federal Member for Calare, Andrew Gee, said the TGA's approval was an important moment for all Australian communities.
"Today's approval is another big step forward for the Central West, particularly in the protection of our most vulnerable people," he said.
"The approval of the Pfizer/BioNTech vaccine is an important step in the fight against COVID-19 and comes after a rigorous and independent assessment, which shows it meets Australian standards for safety, quality and efficacy."
Mr Gee said news of the approval would add further confidence to Australia's vaccine strategy.
"Outside of wartime and the Great Depression, it's hard to think of a worse year for our country than 2020. We've had drought, fires, floods and COVID-19," he said.
"The news of the Pfizer/BioNTech vaccine approval lets us start the year with hope and optimism that there are better times ahead.
READ ALSO:
"Australians should take confidence in the thorough and careful approach taken by the TGA - they have placed the safety of the vaccine above all else.
"Our priority has always been to keep Australians safe and protect lives and livelihoods and that is why our vaccination program is based on expert medical advice, from our medical panel led by Professor Brendan Murphy.
"I would like thank all those involved in the development and assessment of this COVID-19 vaccine, including the researchers, Pfizer, BioNTech and the medical experts at the TGA who have worked around the clock and over Christmas.
Mr Gee said while news of the vaccine's approval came at a vital stage during the fight against COVID-19, communities have been reminded remain vigilant - so if you have any flu-like symptoms, please go and get tested."
The vaccine will be rolled out in five phases over the coming months and, over time, will involve more than 1,000 vaccination administration sites.
As the rollout begins across between 30 to 50 hospital sites, people who need protection the most will get the vaccine first. This includes aged care and disability care residents and workers, frontline health care workers, and quarantine and border workers.
The priority groups have been determined based on the advice of the Australian Technical Advisory Group on Immunisation (ATAGI) which oversees Australia's highly successful immunisation program.

